Wet Age-Related Macular Degeneration
What is Wet Age-Related Macular Degeneration?
Age-related macular degeneration (AMD) is a problem with the retina. It happens when a part of the retina called the macula is damaged. With AMD, the patient loses their central vision, but the patient’s peripheral (side) vision will still be normal.
There are two basic types of macular degeneration: “wet” and “dry.” Approximately 10-15% of the cases of macular degeneration are the “wet” (exudative) type. In the “wet” type of macular degeneration, abnormal blood vessels (known as choroidal neovascularization or CNV) grow under the retina and macula. These new blood vessels may then bleed and leak fluid, causing the macula to bulge or lift up from its normally flat position, thus distorting or destroying central vision. Under these circumstances, vision loss may be rapid and severe.
Age-related Macular Degeneration (AMD) is a leading cause of blindness. There are 20 million people with neovascular macular degeneration worldwide.
Current Treatment
The Standard of Care includes several anti-VEGF-A biologic drugs. The main mechanism of anti-VEGF biologics is to prevent abnormal blood vessel leakage from the choroid to the retina, thereby preventing the formation of retinal fluid and vision loss.
Anti-VEGF-A biologic drugs improve visual acuity and patients’ quality of life, however, they induce no retinal structural vessel change and do not result in disease modification. Some patients have refractive effects on current anti-VEGF therapies or become resistant over time. Despite the monthly treatment with anti-VEGF drugs for two years, 51.5% of the patients treated with ranibizumab and 67.4% of the patients treated with bevacizumab had persistent time-domain optical coherence tomography (OCT) Evidence of liquid.
Not all patients treated with 2.0 mg aflibercept will respond: After one year of treatment, as assessed by angiography or OCT, 19.7%-36.6% of patients have active exudation. An important factor that leads to poor efficacy of anti-VEGF drugs is insufficient dosage, because frequent intravitreal injections make drug absorption poor. All currently prescribed anti-VEGF biologics require intravitreal injection every one or two months, which is a great burden for patients. Studies have shown that in the first year of treatment, although the average patient needs 7 to 8 injections, in fact only 4 to 5 injections. The burden of monthly disease monitoring and multiple intravitreal injections can lead to insufficient doses and unsatisfactory results.
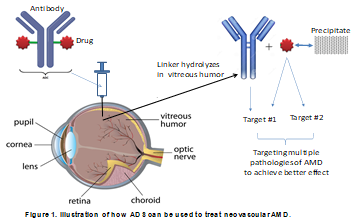
Cloudbreak Solution
Cloudbreak developed a novel way to improve nAMD treatment: antibody-drug synergism (ADS). The idea is to connect an antiangiogenesis antibody to anothor small molecule drug via a linker that can be enzymatically cleaved in the vitreous humor to release the MKI. The small drug targets additional pathological pathways to enhance the effectiveness of the antibody drug.